Speaker
Description
Amyloidosis are systemic diseases characterized by deposition of misfolded proteins into cross-β amyloids in multiple organs in humans and animals. During chronic inflammation, serum amyloid A protein (SAA) levels increases, which result in AA amyloidosis. There is a extreme disease prevalence in captive animals, e.g. 70% in captive cheetah and 57-73% in domestic short hair cats kept in shelters [1], and therefore a prion-like mechanism in amyloid formation has been proposed to explain the horizonal transmission od the disease.
Herein, we present the 3.3 Å resolution cryo-EM structure of fibrils from renal tissue of a cat affected by severe AA amyloidosis in a shelter (Fig. 1) [2]. The fibril is assembled from two twisted proto-filaments, each comprising 76 residues. Amyloid fold and fibril assembly differ from previously reported human and mouse ex vivo AA amyloid structures. Almost identical fibril sequences and similar disease prevalence in related captive cheetah suggest that the structure reported here may depict the prion agent responsible for the high AA amyloidosis prevalence in these two related felids.
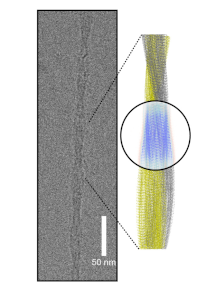
Figure 1. Cryo-EM image of a single fibrilwith the reconstructed map and model
[1] F. Ferri, S. Ferro, F. Porporato, C. Callegari, C. Guglielmetti, M. Mazza, M. Ferrero, C. Crinò, E. Gallo, M. Drigo, L.M. Coppola, G. Gerardi, T. Schulte, S. Ricagno, M. Vogel, F. Storni, M.F. Bachmann, A. Vogt, S. Caminito, G. Mazzini, F. Lavatelli, G. Palladini, G. Merlini, E. Zini. BioRxiv 2022.05.04.490646. 2022
[2] T. Schulte, A. Chaves-Sanjuan, G. Mazzini, V. Speranzini, F. Lavatelli, F. Ferri, C. Palizzotto, M. Mazza, P. Milani, M. Nuvolone, A. Vogt, G. Palladini, G. Merlini, M. Bolognesi, S. Ferro, E. Zini, S. Ricagno. bioRxiv 2022.05.09.491126. 2022

